Breakthrough treatments and clinical trials do not always mean new technology, but entirely new applications of existing technology. The IBD Center at Columbia has enrolled the first patient in a nationwide study using the established technique of sacral nerve stimulation (SNS) in a new way, for the treatment of Crohn’s Disease and ulcerative colitis.
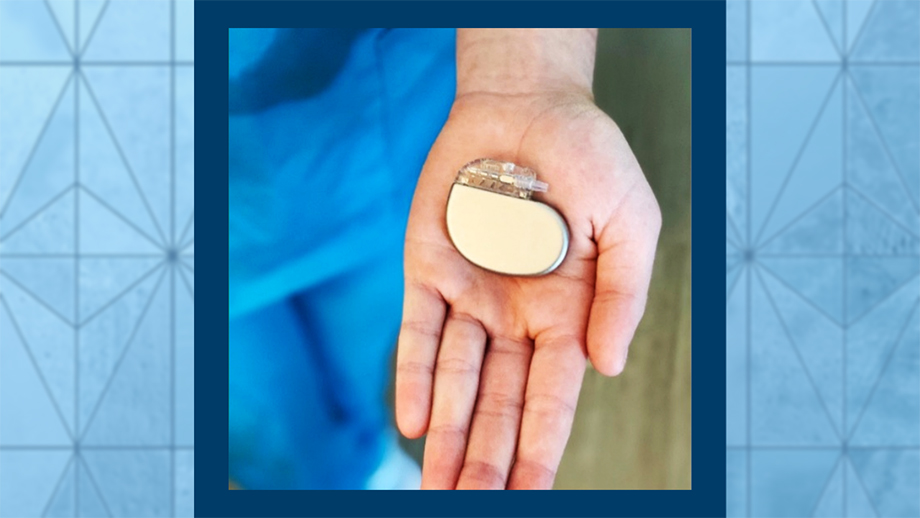
A type of electrical stimulation therapy, SNS uses a small device implanted under the skin at the top of the buttocks to stimulate the sacral nerve with a series of daily electrical pulses. While sacral nerve stimulation has been a routine part of care for fecal incontinence for quite some time, NYP/Columbia is the first institution in the country to offer SNS for inflammatory bowel disease (IBD) as part of the BOOM-IBD trial, an early feasibility study.
We spoke with principal investigator Marco B. Zoccali, MD, FACS, about the importance of this study, trial eligibility, and the ins and outs of sacral nerve stimulation for IBD.
Tell us a little bit about the history of the SNS device and what led to using the device to treat IBD.
It's the same device that has been used since early 2000 here in the U.S. It’s approved by the FDA for fecal incontinence and is also used for chronic constipation in Europe. There have been a few iterations of the device, but it's now been used in over 400,000 patients with a very solid track record in terms of safety and low incidence of complications.
There have been some observations that patients with incontinence who also had IBD would get better. Once they were treated for incontinence with the neuromodulating device, inflammation would improve.
In parallel, there has been a lot of research into the ability of the parasympathetic nervous system to communicate extensively with the intestine. The intestine has the largest number of neurons in our body after our brain. Data shows that by stimulating the vagus nerve in different ways, you can actually modulate inflammation in the bowel. But that’s tricky to do because we would have to access the vagus nerve in the neck and in the chest.
Now we have this device that seems to modulate the impulses of the parasympathetic system to the gut by stimulating the sacral nerve. No need to access the neck, no need to access the chest.
What does stimulating the nerve through the parasympathetic system actually do?
By stimulating a sacral nerve root we can actually trigger the parasympathetic nerves to release impulses that restore balance in the inflammatory versus anti-inflammatory signals in the intestine.
In experimental models, you can actually induce ulcerative colitis in rats pretty reliably. It looks very similar to ulcerative colitis in humans. They would induce ulcerative colitis and then they would treat these animals with sacral neuromodulation. The same thing we would do in humans with the same technique. And invariably all the rats that were treated this way, their intestine would regress.
Within 10 days, they would observe the intestine inflammation by looking at the intestine itself, measuring the cytokines, and also seeing how the rats did. The rats that were treated would survive, and the ones that were not treated would actually get sicker from the disease. Very, very solid experimental data. That’s how the trial started.
Wow. So, that initial research really piqued your interest?
I have always had an interest in this, so I was initially put in touch with the team at Johns Hopkins University doing this study. And I've been doing sacral neuromodulation ever since. The FDA-approved indication [for fecal incontinence] is already life-changing.
Fecal incontinence is a black box. People don't talk about it. It's not very glamorous, and even patients that have the problem don't talk about it. Patients that had the problem solved don't talk about it. Nobody wants to talk about it. But it's life-altering. People stop going out, people don't live their normal life, they get depressed. It’s more prevalent than many other diseases and we don't have anything else to treat this group of patients.
My other interest in colorectal has always been IBD. And as a surgeon of course I love doing surgery, but in IBD we do surgery when everything else fails. And for me, putting the two things together, being able to offer something to this patient before they get to the point of needing a surgical procedure sounded like something that could be potentially revolutionary.
Would the SNS device be something used in tandem with medication or could it potentially replace medication?
That's a very good question. Our understanding of inflammatory disease is getting better and better. It's still not perfect, but the more we learn, the more we understand there are definitely components to the disease.
And again, this is a pilot study. We are enrolling pretty broadly—patients that have been exposed to medication and they're not necessarily going into remission with medications, or even patients that are naive to medication and they would like to explore an alternative that is non-pharmacological. Basically, relying on their own body to reestablish that balance in the intestine.
We don't know exactly if this is going to work for every patient, if it's going to work for both Crohn's disease or ulcerative colitis, in every site in the bowel. The hope is to identify where and for who it works. Ideally, it will work for everybody. But if not, maybe we'll identify a subpopulation that benefits from it. Maybe it will work in conjunction with pharmacological options in some others.
Is the goal to address the inflammation as the primary issue in IBD and then from there it would help with the other symptoms?
We think there is the potential that this could treat the disease, actually tone the inflammation down. We are using the device with a different purpose, not just to manage symptoms.
In a study just published from another institution, they use sacral nerve stimulation via acupuncture. And they showed that 75, 80 percent of the patients with ulcerative colitis that they treated with acupuncture, the ulcerative colitis improved.
The issue with acupuncture is that it's a little bit easier in a patient with a low BMI and you have to go in every day to get it stimulated. But it's the same principle. They manipulate the sacral nerves; they stimulate them with the hope of triggering the parasympathetic system and they have to do it with daily application of the needles.
How does that process differ from implanting the SNS device?
Like a pacemaker for the heart, it's a pacemaker for the gut. We put it in a pocket in the skin and you forget about it. It stimulates an hour a day automatically. The patient has to do nothing, no restrictions, no limitations.
The device is compatible with MRI. It's a one-day procedure on sedation and they go back to their normal activities without any restriction. Again, there are a lot of signals that suggest this could actually deeply shut off the disease, not just mitigate the symptoms that the device has been used for. I think this is a very important concept to stress: This could be a tool that would cure the disease, not just manage the symptoms.
Who is eligible for the trial?
Patients who have active disease — We're enrolling patients with both Crohn's disease and ulcerative colitis between the age of 18 and 75.
They can either be medication naive or if they are on medication, they should be stable on their medication. That judgment is left to the gastroenterologist's discretion whether they've reached the therapeutic levels on their medications. And they can have moderate or severe diseases as long as they don't have severe complications — they shouldn't be in hospital with an acute flare. They are allowed to have had a previous bowel resection.
Many, many patients can actually qualify for this procedure.
What are the primary outcomes you’re hoping to achieve?
We are hoping to achieve remission.
At baseline, we are evaluating the endoscopic grading of the disease. And most of the time biopsies are obtained (that's also left at the discretion of the gastroenterologist). We assess the patient's symptoms, urgency, number of bowel movements, and bleeding in the stool. And also assess a marker of inflammation in the stool like fecal calprotectin and a bunch of inflammatory and anti-inflammatory cytokines in the bloodstream.
Then the patient receives the device implant, and we reassess. We have a two-week follow-up, it's just a questionnaire and wound check, and then a one-, three- and six-month follow-up.
Is the idea that a patient would keep this device for life, doing stimulations every day?
Yes, we’re stimulating the nerve in a permanent way. In the current device, the battery lasts about 15 years and if it needs to be replaced, it’s a same-day procedure. We don't have to replace the wire, we just open the pocket, swap the device, and put in a new one. It takes 20 minutes in the operating room. We know the device can stay there for a long period of time with no side effects.
Are there any risks with this? It really sounds like a dream come true if it works!
Everything has risks, even crossing the street. But again, and I cannot emphasize this enough, the device is not an experiment. It's the same device that I put in a few times a month in patients with fecal incontinence. It's the same thing, the same technique, the same device.
The main risk is that the device gets infected. And if that's the case, the device needs to be removed. Permanent damage to the nerves has pretty much never been seen. But some people can have neuropathic pain. It's described in the single digits and I'm talking about post-market data of hundreds of thousands of patients that have been implanted over time. And if you look, the complication rates are well below 5 percent. Overall, serious complications are really hard to imagine.
As an IBD center, we were leading the way on this with the hope that we can truly make an impact. We are being ambitious, but again, our goal is to actually cure the disease.
Click here to learn more about trial enrollment.
Enrollment Contact:
Call (212) 342-4102
Maeve D Bleistein: mb4971@cumc.columbia.edu
Claudia Musat, MD: cm2065@cumc.columbia.edu
Sign up for our mailing list for more health news and information from Columbia.